 
The packing efficiency of the two – dimensional square unit cell shown below is (A) 39.27 % (B) 68.02 % (C) 74.05 % (D) 78.54 % 48. 
11.3 What is its value for q O 11.4 Give an expression for the unit cell lattice parameter 'a' in terms of penny radius 'r'. 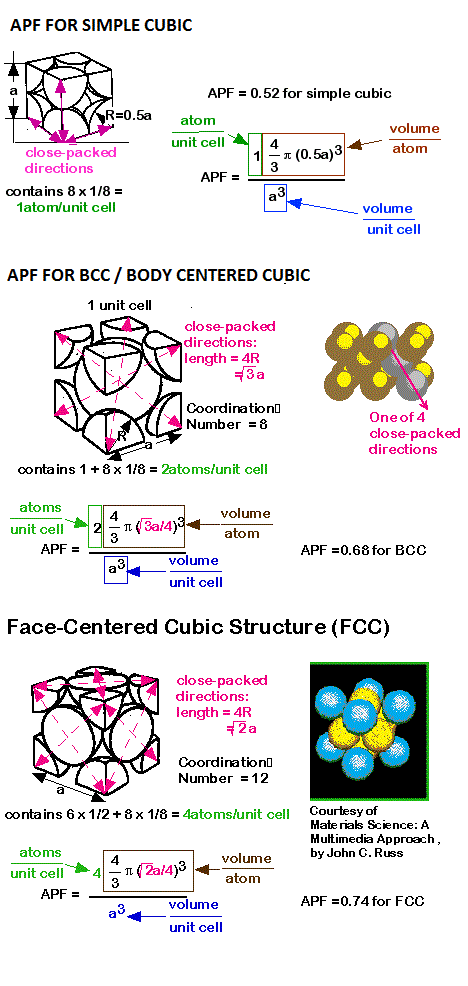
Answer: Molar mass of cscl = 168.5 g /mol Number atoms present in per unit cell for bcc (cscl) Each atom contributes one eight to the unit cell. We chose DNA as the coating material for its high buoyant density (~1.7 g ml −1 in CsCl … Get latest IIT JAM 2021 syllabus PDF at. Let's start with anions packing in simple cubic cells. Finally, we calculate the packing efficiency by dividing the volume occupied by atoms by the volume of the cubic unit cell, a3: Practice Exercise Determine the packing efficiency by calculating the fraction of space occupied by atoms in a body-centered cubic metal. 11.5 Find the packing fraction (i.e., packing efficiency… The Wigner-Seitz (Primitive) Unit Cell for the FCC lattice Thus no. #3- Radius of atom/ion & Packing Efficiency. is the fraction of space in a crystal that is actually occupied by atoms. This arrangement is called ABAB.,… pattern or hexagonal (HCP) close packing. 
No matter how the particles are arranged in the lattice there is always some vacant space inside which we also call voids. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
